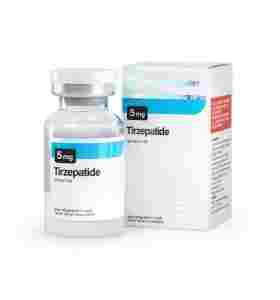
TIRZEPATIDE 5 Deus Medical
🔥 15% EXTRA DISCOUNT FOR DEUS MEDICAL🔥

- Brand: Deus Medical
Shipping
$35 flat rate
Payment Methods
Crypto, Venmo, Zelle, Credit Card, WU
Secure Shopping
Your data is protected with the highest security standards.
⚠️ WARNING: Potential Risks of Anabolic Steroids
Anabolic steroids can have significant effects on the body and should only be used under proper guidance. Misuse or unsupervised use can lead to health complications. These may include hormonal imbalances, liver strain, cardiovascular issues, psychological effects, and more.
If you choose to use anabolic steroids, it is essential to do so responsibly and only obtain them from trusted, verified sources.
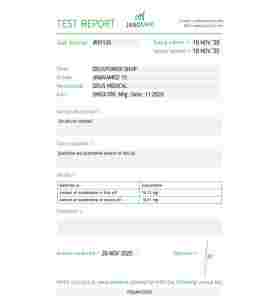
TIRZEPATIDE 5 mg Deus Medical
Exploring the Potential of a Dual-Action Research Peptide
Important Disclaimer: The information provided in this article is strictly for general informational and scientific review purposes. TIRZEPATIDE 5 Deus Medical is discussed here solely as a "research compound." This text does not provide definitive medical advice, dosage instructions, or guarantees of results. Always conduct your own comprehensive research and consult with a licensed healthcare professional or endocrinologist before considering the use of any peptide or research compound.
What is TIRZEPATIDE 5 Deus Medical?
TIRZEPATIDE 5 Deus Medical is a cutting-edge compound currently being investigated as a metabolic modulator. In scientific literature, it is recognized as a first-in-class dual agonist, meaning it is designed to target two separate pathways simultaneously: the GLP-1 (Glucagon-like Peptide-1) and GIP (Glucose-dependent Insulinotropic Polypeptide) receptors.
Provided in a 5 mg lyophilized (freeze-dried) vial, researchers are studying this compound to see if mimicking these two natural metabolic hormones can offer a "double-barrel" approach to supporting weight management and Type 2 Diabetes research. It is hypothesized that this dual action may suppress appetite, help stabilize blood sugar, and potentially support fat loss processes more efficiently than earlier single-receptor peptides.
Potential Core Benefits & Mechanism of Action
Research into Tirzepatide’s mechanism of action focuses on how it might optimize metabolic function and appetite control. The primary areas of investigation include:
Potential for Enhanced Satiety: It is believed that the compound may act on the brain’s hunger centers to increase the feeling of fullness. Researchers suggest this might significantly reduce daily caloric intake without the intense mental strain often associated with traditional dieting.
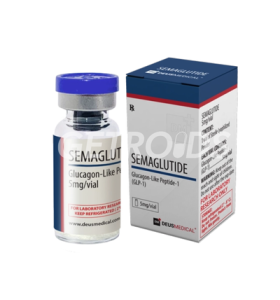
Dual Hormone Signaling: By targeting both GIP and GLP-1 receptors, studies are exploring whether it improves how the body metabolizes sugar and stores fat. This dual action is being compared to GLP-1-only compounds (like Semaglutide) to observe if it yields superior weight management results.
Blood Sugar Regulation: It is hypothesized to stimulate insulin secretion specifically when blood glucose levels are high, while inhibiting excess glucagon release. This mechanism is thought to help maintain stable glucose levels throughout the day.
Delayed Gastric Emptying: Observations indicate it may slow the rate at which food leaves the stomach, potentially keeping test subjects feeling satisfied for longer periods post-meal.
Investigational Weekly Dosing: Due to an extended half-life observed in studies, the compound is typically researched using a once-weekly subcutaneous administration schedule, which is believed to improve overall adherence to the protocol.
Technical Research Specifications
| Feature | Research Specification |
| Manufacturer | Deus Medical |
| Active Substance | Tirzepatide |
| Concentration | 5 mg per Vial |
| Format | Lyophilized Powder (Requires Reconstitution) |
| Classification | GLP-1 and GIP Receptor Agonist |
| Primary Research Focus | Metabolic Weight Management & Glycemic Control |
Observed Research Protocols & Reconstitution
Note: The following metrics reflect standard protocols observed in clinical research settings and should not be taken as medical advice.
In research environments, Tirzepatide is typically introduced on a titration schedule to allow the metabolism time to adjust to hormonal shifts.
Standard Starting Protocol: 2.5 mg per week (observed for the first 4 weeks of studies).
Maintenance Protocol: Up to 5 mg per week (as tolerated by the subject).
Frequency: Once weekly (administered on the same day each week).
Administration Method: Subcutaneous (Sub-Q) application.
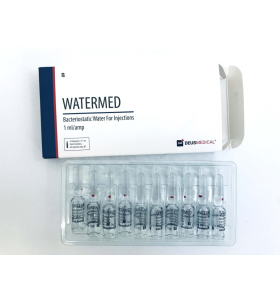
Reconstitution Observations: Researchers typically prepare the 5 mg lyophilized powder by adding 1 ml or 2 ml of Bacteriostatic Water to the vial.
Example: If reconstituted with 1 ml of water, a 2.5 mg research dose corresponds to exactly 50 units on a standard U-100 syringe. The full vial (100 units) would equal the 5 mg dose.
Possible Side Effects & Precautions
Studies note that side effects are generally gastrointestinal and tend to occur most frequently during the initial adaptation weeks:
Gastrointestinal Responses: Nausea and diarrhea are the most commonly reported effects as the digestive system's pace slows down.
Appetite Reduction: While this is a primary focus of the research, the loss of appetite can be profound. In studies, prioritizing protein intake is often emphasized to help maintain lean muscle mass.
Hydration Needs: Because reduced food intake often leads to a decrease in fluid consumption, maintaining adequate hydration is considered critical during research protocols.
Critical Precautions: Subjects with a personal or family history of pancreatitis or thyroid issues are typically excluded from these studies or monitored under extremely strict medical supervision.
Frequently Asked Questions (FAQ)
1. How is Tirzepatide theorized to differ from Semaglutide?
While Semaglutide focuses solely on targeting the GLP-1 receptor, Tirzepatide is designed to target both GLP-1 and GIP receptors. Preliminary clinical data suggests that the addition of the GIP mechanism might allow for a broader metabolic impact, potentially leading to enhanced weight loss and better blood sugar stabilization.
2. Is Tirzepatide being researched for weight loss in non-diabetic subjects?
Yes. Although originally developed and studied for Type 2 Diabetes, it is being heavily researched (and widely discussed off-label) for chronic weight management in overweight or obese individuals, largely due to its potent potential for appetite suppression.
3. Does this compound require a specific diet during research?
Observations suggest that Tirzepatide might make adhering to a calorie deficit easier by reducing hunger cues. However, researchers generally note that for optimal body composition results, the protocol should be paired with a high-protein diet and resistance training to encourage the body to target fat stores rather than muscle tissue.
4. How long does a 5 mg vial last in standard research protocols?
Based on standard study guidelines, if a subject is on the starting dose of 2.5 mg once per week, a single 5 mg vial will last for two weeks. If the subject has titrated up to the 5 mg maintenance dose once per week, the vial serves as a single weekly dose.





















 Ask us anything on Telegram!
Ask us anything on Telegram!